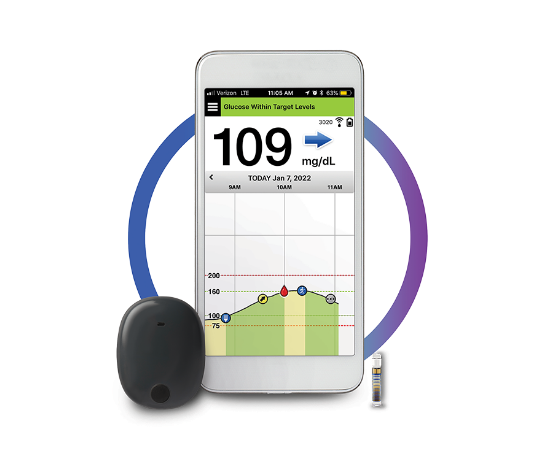
ASCENSIA DIABETES CARE LAUNCHES THE 6 MONTH EVERSENSE® E3 CONTINUOUS GLUCOSE MONITORING SYSTEM IN THE U.S. WITH THE NEW EVERSENSE PASS SAVINGS PROGRAM
Senseonics Holdings Is A Winner With The Ascensia Deal In Glucose Monitoring (NYSE:SENS) | Seeking Alpha

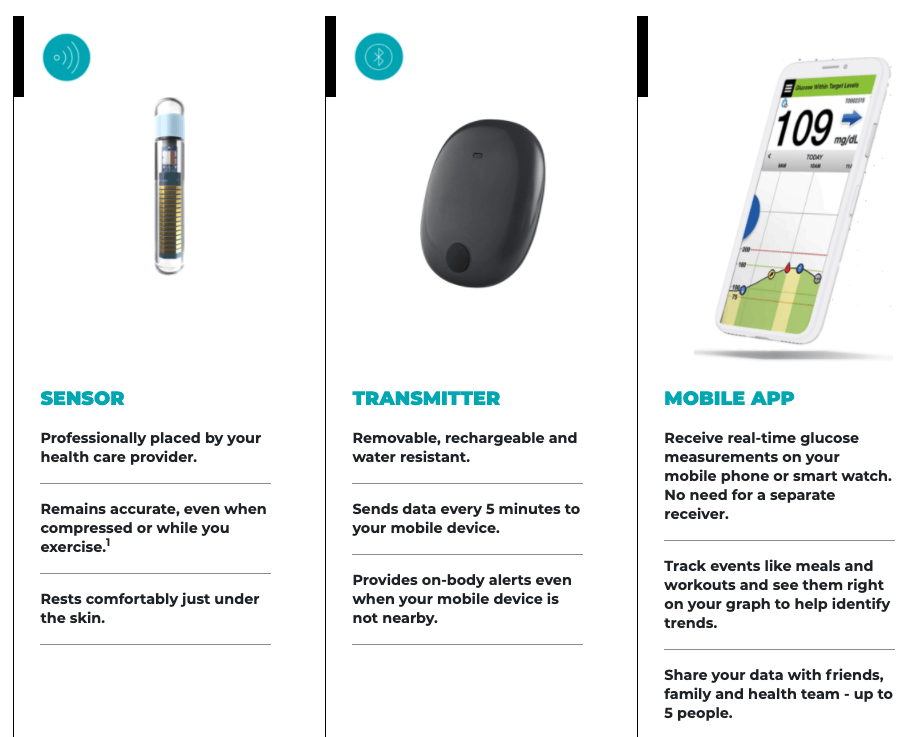
Eversense E3 CGM Approved for Two Sensors per Year: Your “Happily Ever(sense) After” - Taking Control Of Your Diabetes®

Ascensia Diabetes Care on Twitter: "#ICYMI the FDA recently approved the Eversense E3 #CGM system for use for up to six months, offering people with #diabetes more choices for their self-management. Learn

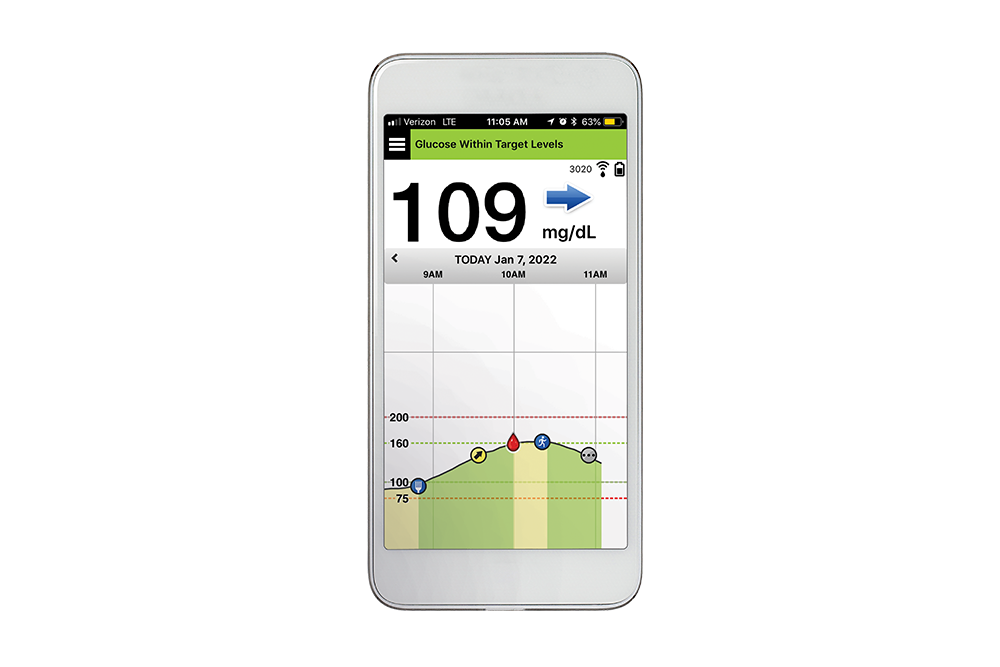











![6-Month Glucose Implant Sensor Receives CE Mark Approval [video]|Health Tech Insider 6-Month Glucose Implant Sensor Receives CE Mark Approval [video]|Health Tech Insider](https://i0.wp.com/healthtechinsider.com/wp-content/uploads/Eversense_CGM_with_video_600x275.jpg?fit=600%2C276&ssl=1)